Trigeminal Nervous System Segmentation
Let us start from a simple consideration: the interpretation of encrypted signals from neural activities is a complex and brave task. However, by implementing a simplification strategy, we can deduce interesting data that might help us understand the ‘Emergent Behaviour’ of the system itself.
We already explained that indeterminated systems should be stimulated by an external trigger in order to be studied. It means that we also have to be aware of the nature of the stimulus: it has to be known in advance and it has to be well calibrated, because a sub-threshold stimulation could generate a type of response from the CNS, while another supra-threshold response would go to a saturated response.
Segmenting the part of the CNS involved in the masticating function results in dividing it into three macro areas:
- the cortical area (in which the response of the trigeminal motor action occurs - i.e. the Trigeminal Cortical Area);
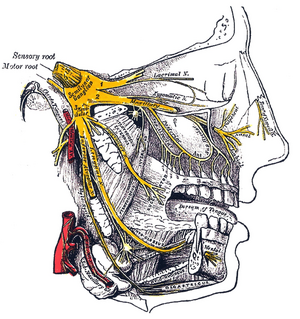
- the peripheral area, where the response of the trigeminal root is analysed; and
- the brainstem-pontine-related area, which is involved in a broad genesis of trigeminal reflexes.
Trigeminal Cortical Area
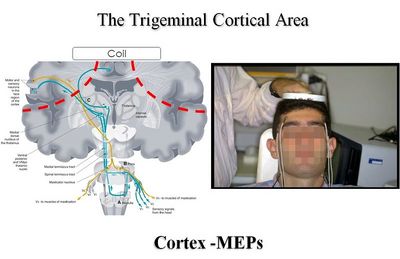
When we try to assess the integrity and/or symmetry of the Trigeminal Motor Cortex (TMC), we certainly cannot completely trust the MR imaging techniques because they lack data about ‘functionality’: the image symmetry of the trigeminal motor cortical areas does not results in functional symmetry, even if we could try a kind of comparison with the functional MRI (fMR). This latest technology, however, is unlikely to be extemporaneous, because the speed of activation of neuronal excitability is too high. As a result, we are forced to employ electrophysiological stimulation techniques to evoke a response from the pyramidal bundle coming from neurons of the first order pf the TMC (Figure 1).
Nowadays electrical transcranial stimulation is no longer used; it has been replaced by the Magnetic Transcranial Stimulation or Stimulation mTCS.
Magnetic Transcranial Stimulation (mTCS)
In our laboratories, this procedure requires a unique experiment in which the electrodes are positioned on the patient and a series of tests is carried out, including:
- the magnetic Transcranial Stimulation mTCS,
- trigeminal reflexes, and
- the Electric Transcranial Stimulation of the trigeminal Root eTCS, as described below.
In Figure 10, we can understand the genesis of the mTCS and subsequent neuromotor responses named Cortex-Motor Evoked Potentials (C-MEPs) elicited by an induced current within the brain tissue; this current was determined by the magnetic field crossed from a coil placed on the subject’s skull vertex. As a routine, we use a Magstim BiStim2 magnetic stimulator (Magstim Ltd, UK), attached to a circular coil (external diameter 10 cm), for magnetic fields up to 2 T (exit 100%). The intensity of stimulation is expressed as a percentage of the maximum power. The magnetic coil is positioned flat on the scalp; the optimum position is on the medial line, with the centre of the coil slightly forward to the top. This position is capable of evoking a motor action potential on both the masseter muscle, without evident depolarization of the trigeminal root. In order to evoke a Cortex-MEPs, subjects need to exercise a mild mandibular closure with EMG activity. The latency at the onset and the peak-to-peak width are measured at least on six tests[1].
Trigeminal Nervous System excitability
Transcranial magnetic stimulation activates the primary motor cortex by excitating the interneural fibres that project on pyramidal cells; it also evocates short series of potential of action called Indirect or I-waves. Magnetic high-intensity shocks can also depolarize the assonal portion of the pyramidal cells stimulating mixed events, including a direct D-wave action potential and some Indirect waves (I-wave)[2].
The stimulation of the facial motor cortex evokes a descendant burst; this burst travels along the corticobulbar tract and reaches the trigeminal and facial motorneurons with a multi-synaptic connection, whereas there is a monosynaptic and almost completely controlateral connection at the masseteric motorneurons that is similar to the corticospinal projection on motor neurons of the hand muscles. Pre-innervation has also to be acknowledged when we have to deal with the trigeminal system: even with high intensity magnetic stimulation, no motor potential can be evoked without contracting the target muscles. During the contraction, potential masseterics show shorter latency, short duration, and synchronous responses, which reach an amplitude of approximately 30% of the motor response from direct stimulation of the masseter nerve named M-wave.
Motoneuronal activation follows the principle of the section: the smaller motorneurons are activated first[2]. Masseteric motorneurons show normal excitability when they are triggered by the stimulation from the motor cortex. Schwartz and Lund (1995) have recently studied the effect of nociceptive pressure on the mandibular movement and the EMG activity of the masseter in decerebrated rabbits. The motoneurons stimulated the corticobulbar tract through the same path, but the two entrances differed. In our experiments the short-lived high frequency discharge of action potential is of a phasic type, even if in the works of Schwartz and Lund[3], the potentials for action are tonic. In their experiments on tonic pain, the amplitude of mandibular movements and the recruitment of the masseteric motorneurons decrease. However, we can exclude a greater excitability all along the path from the motor cortex to the lower motorneurons.
Trigeminal Peripheral Area
When we need to assess the integrity of the trigeminal roots, we should evoke a motor action potential of the peripheral trigeminal motor tract in a synchronous and symmetrical way. In this situation, a m TCS is not suggested: it would rather be advisable to adopt the bilateral electric stimulation routines called bilaterallyRoot-MEPs technology that we fine-tuned in our neurophysiology laboratories[4].
In order to achieve this target, synchronicity, symmetry, and the pulse maximum power are essential. This is why electromyographers must be used for evoked potential and they must be equipped with two independent and autonomous high voltage electrostimulators. The EMGs that employ tools with a single multiplexated electrostimulator to deliver two or more stimuli are not appropriated: the second impulse would lose synchronicit, because the condensers need time to recharge (Figure 2).
We carry out an analysis made through a generic finite element processor (FE, SimNibs method) to study the distribution of the electric field within the intracranial brain tissue, even if only as a descriptive model (Figure 3)[5].
The EF models of the study consist of about 1.7 million tetrahedra. The Mesh resolution was selectively improved in the regions of the Grey Matter (GM), White Matter (WM), skull, and cerebrospinal fluid (CSF), with an average tetrahedron volume of 1 mm3.
Electrical conductibility has been assigned to different types of the affected tissues in which
[6].
Figure 3A shows the position of the electrodes (Figure 12A); the maximum current will spread under the cathodes (red in colour) in the parietal cortex (Figure 3B); in the cranial cortex region, near the trigeminal motor root, the current density is of low magnitude (Figure 3C, black arrows). Figure 3D shows the current density that spreads in the brain tissue. It underlines the least amount of electrical current within the brain tissue necessary to saturate the trigeminal motor (relative to the amount required to evoke a response of the trigeminal motor cortex under the cathode). This is one of the reasons that urged us to choose this kind of evoked response (absolutely peripheral) rather than the cortical one (with higher threshold, lower stability, and less focus).
With this approach we concluded that, given the emergence of the trigeminal root in oval foramen and the distance from the parietal bone and parietal cortical area, the cathode positioning should be more caudal than the regulation (11–13 cm on the acoustic-meacoustic line): immediately above the upper edge of the ear. The position of the cathode, therefore, will be approximately more than the starndard 13–15 cm and will be delimited by the upper margin of the ear.
Considering the safety limits, the energy provided for each individual impulse in our application will follow this formula:
per impuls.
About the simultaneous use of the two electrostimulators — from the safety point of view — the limits turn out to have been 10 times lower than those indicated in the IEC legislation.
The electrodes are arranged as follows:
- Cathode electrode has been placed on each side at 13–15 cm along the line conjoining the vertex - acoustic meatus in the bilaterally parietal region.
- The electrical stimulus consists of a square wave which length is 0.250 ms, at a voltage of 300 V and a maximum current of 100 mA.
Hence, the first procedure is to check the saturation of the motor response of the trigeminal root.
- At 20 mA, 30 mA, and 40 mA, we can observe a latency of 2.4 ms, 2.4 ms, and 2.3 ms respectively, but on increasing the amperage, we can observe a decrease in latency up to 2.1 ms at 50 mA, 2 ms at 70 mA, and 1.9 ms at 80 mA, 90 mA, and 100 mA (Figure 3).
- These latency differences reach up to a maximum current density, depending on the capacitive components and resistance of the tissues to the current flow.
The saturation of the electrophysiological signal evoked by the trigeminal root is the first absolute and mandatory step that needs to be performed, even before clinical interpretation. The saturation of the evoked potentials from the trigeminal root showed no change in amplitude. Matter of fact, when the electrostimulator reaches 80 mA pulses, 90 mA, and 100 mA, the P-P amplitude stabilizes at 4.6 mV (Figure 4). The amplitude value of 4.6 mV (of course, the amplitude can be chosen to the integral area or vice versa, depending on the purpose of the study) is to be considered the ‘Maximum Absolute value of Neural Evoked Energy’ from the trigeminal motor system. It is called ‘mANEE’.
Trigeminal Brainstem Area
The trigeminal brainstem area is the most complex area to study and interpret because of the complexity of its multi-synaptic connections. The following electro-physiological tests are objectively sufficient to understand the cryptic language of the SCNS. These will be treated in this section, but they will also be resumed in other editions. The following trigeminal reflexes will be considered: the jaw jerk, the mechanical and electrical silent period, the recovery cycle of the masseteric inhibitory reflex and the laser silent period, as well as the masseteric laser-evoked potentials (Figure 5).
Jaw jerk Reflex
The piezoelectric trigger is used for the mandibular reflex, though it does not provide controlled reproducibility and quantification of the stimulation intensity; simultaneous recordings of the two sides are considered an essential method for the accurate and acceptable assessment of the asymmetry of the side. Asymmetry in the latency is very small: it ranges from 0 to 0.8 ms with an average of 0.13 ms (SD 0.17) in 131 normal subjects[7]. Although in trigeminal neuropathy or multiple sclerosis the jaw jerk reflex may be retarded by several milliseconds, asymmetries of latency of only 0.8 ms were considered to be a higher limit of normality in neurological studies (Figure 6).
In previous studies about the jaw jerk in patients with cranial-mandibular disorders (TMDs), patients with unilateral disorders were selected to identify an affection side in which there was a latency delay and a lower amplitude on the side of the mandibular deviation and pain. Even if the maximum mandibular closure force in TMDs can be reduced to half as much as control groups, several researchers propose that muscular hyperactivity is one of the key mechanisms in cranial-mandibular dysfunction, and that hyperactivity of the masticatory muscles is central to the nervous system[8]. A cortical activity or abnormal reticular system would increase the excitability of the motorneurons directly through the corticobulbar system or indirectly through the modulation of multi-synaptic reflexes of lateral reticular formation. The initial causes could be stress, psychogenic factors, or a primary disease of the central nervous system, such as the oromandibular dystonia[9][10].
The afferents from group II in the division of the maxillary and mandibular trigeminal nerve apply a powerful inhibition of the motoneurons of the mastication muscles through synaptic and polysynaptic reflexes. A remarkable feature of the mandibular reflexes, however, is their bilateral symmetry. In some patients with multiple sclerosis (MS) the latency is prolonged, whereas it lacks in others: this reflex can sometimes be essential for diagnosing the lesions of the brainstem in MS. It might also be more effective for therapy (Figure 6).
Masseteric Mechanical Silent Period
The jaw jerk is a short-latency excitatory reflection that can be evoked by a stretch of the mandibular elevators through a percussion produced by a triggered neurological hammer. The excitation on motoneurons α from the neuromuscular spindles is the only generally accepted explanation. When this type of mechanical stimulus is applied during voluntary activation EMG (i.e., by pressing the teeth), the jaw overlaps with the Interference EMG activity, and is followed by a period of absence or depression of the electromyographic activity, the so-called Masseteric Silent Period (MSP)[11] (Figure 7).
The MSP has sparked particular interest as it has been shown that the duration of the silent period is higher in patients with TMDs[12][13]. This period of depression or absence of EMG activity can be caused by various active inhibitory influences, but it can also be caused by synchronization of the a motoneurons after the initial efferent volley.
The criteria for clinical evaluation remain unclear: there is still a certain confusion about both measurement and reported values, which vary considerably in different studies. Klineberg and collaborators provoked a percussion on the chin through the use of a standardized solenoid in 10 healthy subjects during mass isometric activity: it generated a vertical movement that was parallel to the surface fibres of the superficial muscle of the masseter muscle, and they after analysed the mechanical silent period. The reflection evoked the classic jaw jerk in short latency (mean, 8.3 ms) followed by an SP (average latency, 15.9 ms, average duration, 42.6 ms). The duration of the SP greatly varies not only between subjects, but also within individuals[14].
Recovery Cycle of Masseteric Inhibitory Reflex
Headache is oftes associated with a ‘sensitization’ of the nociceptive trigeminal system with the involvement of anti-nocicective mesenchephalic structures such as periaqueductal substance, locus coeruleus, and the nuclei of the raphe, which have a modulator effect on trigeminal sensitive nuclei[15].
The descending modulator effect from these nuclei has been shown through experimental studies on animals, and the temporary trigeminal ‘sensitization’ effect during hemicranic attack has been shown to be clinically demonstrated through electrophysiological studies[16]. The most credited hypothesis considers the dysfunction of anti-nocicadjective nuclei as the cause of a disinhibition of the trigeminal nociceptive system[17].
Orofacial pain, including algies from TMDs, appears to have a modulation effect on the mandibular reflexes. Electrophysiological studies have shown that the experimentally induced pain by infusion of saline hypertonic solution to 5% in the masseter results in an increase in the peak-to-peak size of the jaw spider. This facilitatory effect seems to be related to the increased sensitivity of the fusimotor system, which causes muscle stiffness[18].
Other animal studies have been carried out in the experimentally induced muscle pain, demonstrating that the activation of muscle nociceptors markedly influences the proprioceptive properties of neuromuscular fusis through a central neural pathway.[19] However, the washing of the local algogena substance leads to a return to normality of the tendon reflexes. In pain from TMDs, there is a dichotomy between schools of thought: while one school proposes an ‘hyperactivity’ effect of the central nervous system (CNS: Central Nervous System), the other suggests a normal excitability of the trigeminal nervous system[20][21].
Bruxism is usually believed to be related to musculoskeletal pain, such as pain from TMDs, but also to muscular-tensive headache. Few studies have actually tried to fully describe the pain associated with bruxism examining the neurobiological and physiological characteristics of the mandibular muscles. There are some clinical cases in the literature and small studies suggesting that some drugs related to dopaminergic, serotonergic, and adrenergic systems, can both suppress and exacerbate bruxism. Most of the pharmacological studies mentioned before indicate that several classes of medications can affect muscle activity related to bruxism without having any effect on Orofacial Pain (OP: Orofacial pain)[22]. It seems that the ‘sensitization’ of the trigeminal nociception system, the facilitation effect on the mandibular reflesxs, and the hyper-excitability of CNS are neurophysiopathogenetic phenomena that are related to pain in the craniofacial district. At the same time, the recovery cycle of the torpedo period shows the level of neuronal excitability of the trigeminal system, and it could be a valid method of testing the excitability of the nervous system.
The recovery cycle of the masseteric reflection reflex (rc MIR: recovery cycle Masseteric Inhibitory Reflex) has been studied by generating pairs of stimuli with identical characteristics provided percutaneously by an electrical stimulator positioned on the patient’s face in the emergency area of the mental nerves (Figure 8).
Stimulation was carried out with a square-wave electrical stimulation that was 2.5 times the inhibitory reflection threshold, which was able to evoke a distinct rc MIR, composed of the two silent periods called ‘exteroceptive suppression’ to distinguish them from separate mechanical Silent Periods (SPs); these can be recognized in a first silent period called ES1 (Exteroceptive Suppression 1) and a second silent period named ES2 (Exteroceptive Suppression 2) interspersed by the resumption of the activity EMG (Interposed IA Activity). The first stimulus (S1) is considered to be conditioning and the second (S2) a stimulation test. The interstimulus range to verify the presence of a neural hyperexcitability between S1 and S2 has been set at 150 ms.
In our studies, healthy subjects were instructed to tighten their teeth to produce the maximum EMG activity and maintain it for at least 3 seconds with the help of visual and sound feedback. After 60 seconds of rest, the subject repeated the contraction. The EMG signal was recorded both in direct mode and in mediated mode. The disposition of the recorder electrodes must be the same as for the registration of the b Root, jaw jerk, and SP. The parameters of the preamplifier will have to be set in 50 ms per division, 100 mV per division, and the filter bandwidth of 50 Hz–1 kHz.
Laser Evoked Potentials and Masseteric Laser Silent Period
To evoke motor responses from the masticatory muscles or potential laser evenings, we employ a laser stimulator CO2 (Neurolas, Florence, Italy) capable of generating a radiant caloria (10.6 mm; intensity 1.5–15 W; duration of 10–15 ms; diameter of the 2.5 mm spot) in the area of the skin in upper and lower upper and lower region (trigeminal terrain V2 and V3). The subjects must sit comfortably in the dental chair and wear protective goggles (Figure 9). To avoid the habit to the nociceptive stimuli and the overheating of the skin, the irradiated points have been shifted after each stimulation.
The perception threshold has been determined by the limits method in two sets of intensity by increasing or decreasing the trigger stimulation. The intensity of the laser beam is fixed at 1.5 the perceptual threshold. With regard to LEP (LEP: Laser Evoked Potentials), signals are recorded through the top-disk electrodes (Cz) with two references for each side on the ear lobes (A1, A2). Two blocks of eight to 12 tests each are mediated off-line. Signs are amplified, filtered (0.5–50 Hz), and stored by means of an analyser for biopotential (Premiere, Medelec, UK). For each block, we have measured the latency of the N and P components and the peak-to-peak width of the potential evoked.[23]
For LSPs, the same parameters were used as described above, but the recording is performed on masseter muscles. The area was subtracted from the 100-ms curve prior to adjusted and mediated laser stimulus (pre-analysis). The duration of the registered EMG activity is 400 ms, of which 100 ms corresponds to pre-stimulus and 300 ms in the post-stimulus period. The EMG signals are amplified, filtered (20 Hz–1 kHz), and sampled at 4 kHz. Subjects are asked to tighten their teeth with the maximum muscular strength to determine the EMG activity corresponding to the Maximum Volunteer Contraction (MVC: Maximum Voluntary Contraction) of masseter muscles and at different levels (15–25%), (35–45%), (55-65%), and (75–85%) of MVC. Subjects receive visual feedback with markers on the computer screen, which clearly indicates when the default level is reached.
As Figure 10 shows, the results of the following work are schematized in accordance with the type of test performed. With a laser stimulation in the skin region corresponding to the emergence of the supraorbital nerve (V1), we can have a Blink Reflex as a reflected response (BR) and notice the perfect symmetry of the responses on sides R1 and R2. Stimulation in the perioral region will result in a reflex response from the masseterini muscles called Laser Silent Period (LSP: Laser Silent Period). We might note, in this test, a slight asymmetry of the track, mainly caused by the different degree of motoneural recruitment in the maximum intercuspidation. The registration on the scalp determines the potential laser summers of the trigeminal somatosensory area (LEPs) and measure the negative and positive spikes (N and P). Here too, we witness a high level of symmetry.
The Exposed Laser procedure is very interesting because — as shown in Figure 19 — there is evidence of a high symmetry of the blue component of the Blink reflex (R2 right and left) that corresponds to the motor nerve activity of the facial nerve; a relative somatosensory symmetry might be detected (N2 and P2, while a clear asymmetry of the MCV masseterin (MIR) is denoted in the width of the motor unit by both stimulus and post-inhibition. This introduces an important concept of the neural symmetry that might reveal an extraordinarily fascinating world of neurophysiopathological notions in the field of mastication.
- ↑ Cruccu G, Berardelli A, Inghilleri M, Manfredi M, «Functional organization of the trigeminal motor system in man. A neurophysiological study», in Brain, 1989».
DOI:10.1093/brain/112.5.1333 - ↑ 2.0 2.1 Rothwell JC, Thompson PD, Day BL, Boyd S, Marsden CD, «Stimulation of the human motor cortex through the scalp», in Exp Physiol, 1991».
DOI:10.1113/expphysiol.1991.sp003485 - ↑ Schwartz G, Lund JP, «Modification of rhythmical jaw movements by noxious pressure applied to the periosteum of the zygoma in decerebrate rabbits», in Pain, 1995».
DOI:10.1016/0304-3959(95)00028-q - ↑ Frisardi G, «The use of transcranial stimulation in the fabrication of an occlusal splint», in J Prosthet Dent, 1992.».
DOI:10.1016/0022-3913(92)90345-b - ↑ Windhoff M, Opitz A, Thielscher A, «Electric field calculations in brain stimulation based on finite elements: An optimized processing pipeline for the generation and usage of accurate individual head models», in Hum Brain Mapp, 2013».
DOI:10.1002/hbm.21479 - ↑ Thielscher A, Opitz A, Windhoff M, «Impact of the gyral geometry on the electric field induced by transcranial magnetic stimulation», in Neuroimage, 2011».
DOI:10.1016/j.neuroimage.2010.07.061 - ↑ Kimura J, Daube J, Burke D, Hallett M, Cruccu G, Ongerboer de Visser BW, Yanagisawa N, Shimamura M, Rothwell J, «Human reflexes and late responses. Report of an IFCN committee», in Electroencephalogr Clin Neurophysiol, 1994».
DOI:10.1016/0013-4694(94)90131-7 - ↑ Molin C, «Vertical isometric muscle forces of the mandible. A comparative study of subjects with and without manifest mandibular pain dysfunction syndrome», in Acta Odontol Scand, 1972».
DOI:10.3109/00016357209002499 - ↑ Yemm R, «Neurophysiologic studies of temporomandibular joint dysfunction», in Oral Sci Rev, 1976».
- ↑ Cruccu G, Pauletti G, Agostino R, Berardelli A, Manfredi M, «Masseter inhibitory reflex in movement disorders. Huntington's chorea, Parkinson's disease, dystonia, and unilateral masticatory spasm», in Electroencephalogr Clin Neurophysiol, 1991».
- ↑ Goldberg LJ, «Masseter muscle excitation induced by stimulation of periodontal and gingival receptors in man», in Brain Res, 1971».
DOI:10.1016/0006-8993(71)90330-1 - ↑ Widmalm SE, «The silent period in the masseter muscle of patients with TMJ dysfunction», in Acta Odontol Scand, 1976».
DOI:10.3109/00016357609026557 - ↑ Brenman HS, Black MA, Coslet JG, «Interrelationship between the electromyographic silent period and dental occlusion», in J Dent Res, 1968».
DOI:10.1177/00220345680470033301 - ↑ Hellsing G, Klineberg I, «The masseter muscle: the silent period and its clinical implications», in J Prosthet Dent, University of Sydney, Faculty of Dentistry, 1983, Sydney, New South Wales, Australia».
DOI:10.1016/0022-3913(83)90249-4 - ↑ Holle D, Obermann M, Katsarava Z, «The electrophysiology of cluster headache», in Curr Pain Headache Rep, 2009».
DOI:10.1007/s11916-009-0026-9 - ↑ Yan J, Melemedjian OK, Price TJ, Dussor G, «Sensitization of dural afferents underlies migraine-related behavior following meningeal application of interleukin-6 (IL-6)», in Mol Pain, 2012».
DOI:10.1186/1744-8069-8-6 - ↑ Hansen PO, Svensson P, Arendt-Nielsen L, Jensen TS, «Human masseter inhibitory reflexes evoked by repetitive electrical stimulation», in Clin Neurophysiol, 2002».
DOI:10.1016/s1388-2457(01)00729-5 - ↑ Truini A, Romaniello A, Svensson P, Galeotti F, Graven-Nielsen T, Wang K, Cruccu G, Arendt-Nielsen L, «Experimental skin pain and muscle pain induce distinct changes in human trigeminal motoneuronal excitability», in Exp Brain Res, 2006».
DOI:10.1007/s00221-006-0508-5 - ↑ Louca S, Christidis N, Ghafouri B, Gerdle B, Svensson P, List T, Ernberg M, «Serotonin, glutamate and glycerol are released after the injection of hypertonic saline into human masseter muscles – a microdialysis study», in J Headache Pain, 2014».
This is an Open Access resource! - ↑ Xu W, Wu Y, Bi Y, Tan L, Gan Y, Wang K, «Activation of voltage-gated KCNQ/Kv7 channels by anticonvulsant retigabine attenuates mechanical allodynia of inflammatory temporomandibular joint in rats», in Mol Pain, 2010».
DOI:10.1186/1744-8069-6-49
This is an Open Access resource! - ↑ Cruccu G, Frisardi G, van Steenberghe D, «Side asymmetry of the jaw jerk in human craniomandibular dysfunction», in Arch Oral Biol, 1992».
DOI:10.1016/0003-9969(92)90047-c - ↑ Winocur E, Gavish A, Voikovitch M, Emodi-Perlman A, Eli I, «Drugs and bruxism: a critical review», in J Orofac Pain, Tel Aviv University, 2003, Tel Aviv, Israel».
- ↑ Romaniello A, Cruccu G, Frisardi G, Arendt-Nielsen L, Svensson P, «Assessment of nociceptive trigeminal pathways by laser-evoked potentials and laser silent periods in patients with painful temporomandibular disorders», in Pain, 2003».
PMID:12749956
DOI:10.1016/s0304-3959(02)00347-0
particularly focusing on the field of the neurophysiology of the masticatory system